IDSA updates COVID guidelines for antibodies, antivirals, and other medications
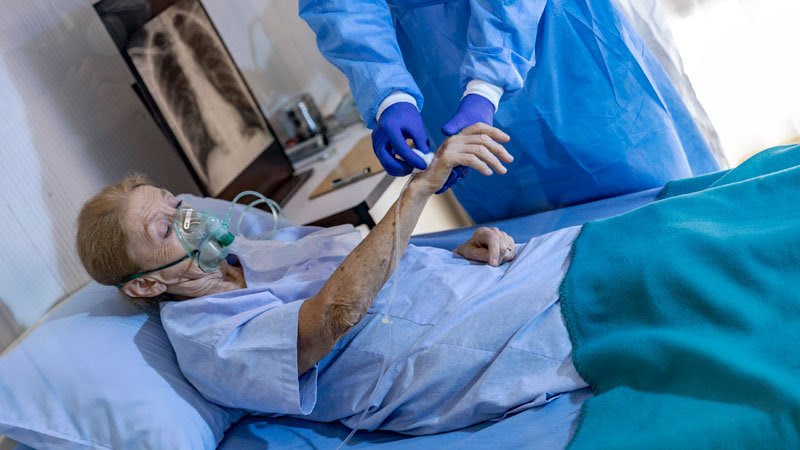
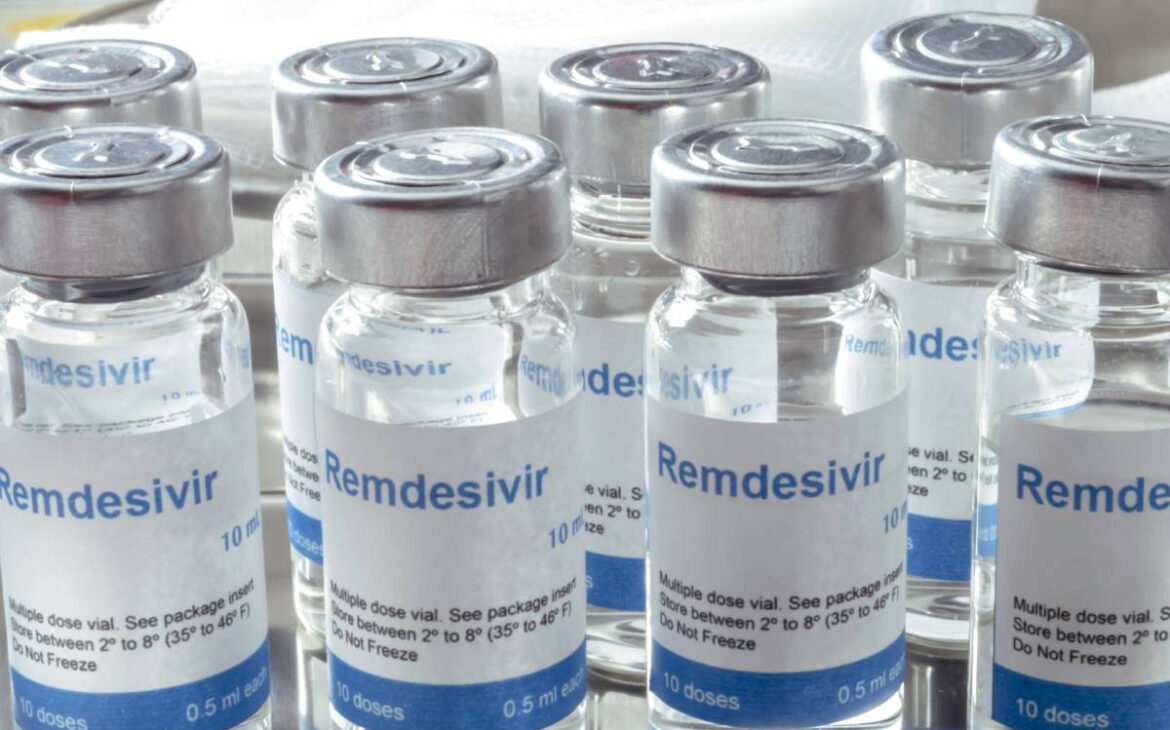
Por Damian McNamar Un panel de expertos en enfermedades infecciosas advierte contra el uso rutinario de bamlanivimab (Eli Lilly) y señala que el remdesivir (Veklury) puede acortar el curso clínico de COVID-19, que podría ser crítico «a medida que los hospitales se llenen» en los Estados Unidos. El grupo también dijo que los anticuerpos monoclonales […]
IDSA updates COVID guidelines for antibodies, antivirals, and other medications Read More »