By John Jesitus
Controlling the first major mpox outbreak in North America required early, sustained, and culturally sensitive engagement with affected communities, along with a timely shift to pre-exposure prevention (PrEP) for those at risk, according to a report published today in Annals of Internal Medicine.
Starting with a handful of cases reported in early May, Montreal Public Health officials had recorded 402 cases of mpox (346 confirmed, 56 probable) by October 18, mostly among men who have sex with men (MSM), and almost all spread through sexual contact.
The document reminds clinicians that current cases of mpox (historically referred to as monkeypox) may not reflect textbook descriptions of clinical manifestations and transmission routes, said co-author Donald C. Vinh, MD, a clinical scientist in infectious diseases at McGill University Health Centre in Montreal, Canada. Perhaps most importantly, he told Medscape Medical News, the Montreal experience underscores the value of partnership in formulating and implementing policies and procedures to mitigate transmission.
“The most important step was philosophical rather than practical,” Vinh said. “And that was a partnership between public health, the doctors on the ground who were encountering patients with this ‘unknown disease’ in the clinic, and the at-risk community.”
Spreading the word
Cooperation was essential in disseminating the message about what mpox is, how it is transmitted, and how to use the appropriate medical and demographic channels to educate people at risk. “You can have an epidemiological definition,” she said, “but there’s also a practical application: it may have been circulating primarily, at least in our outbreak, among men who have sex with men (MSM). That influenced the vaccination strategy, so it wasn’t just people who were engaging in high-risk sexual activity. It was also people who anticipated engaging in activities that could put them at risk, or who identified themselves as being at risk.”
Dialogue between public health officials and the at-risk community revealed that previously unexposed MSM might choose to attend gatherings where they could be exposed. Social venues offering anonymous sex on-site initially accounted for up to 60% of the cases.
Vinh says doctors and policymakers appreciated the community members' openness about their potential risks. "Instead of stigmatizing or punishing them or trying to be condescendingly educational about it," he said, "let's be pragmatic and vaccinate these people who admit to being at risk."
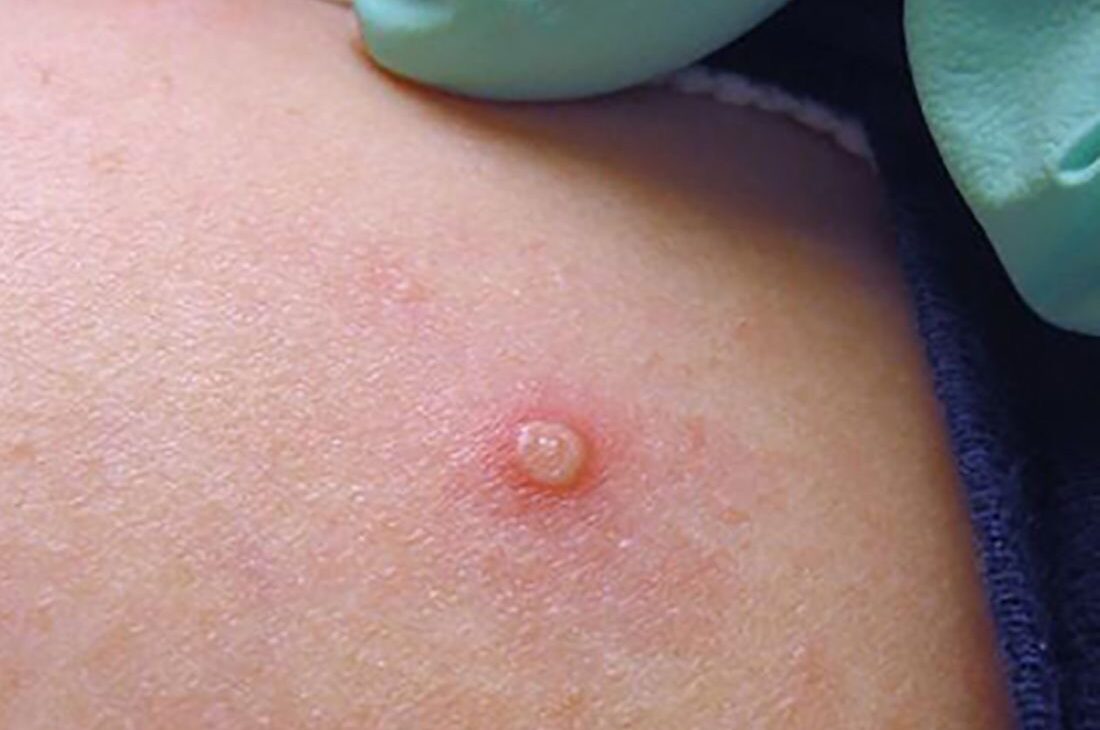
Epidemiological data reflect a rapid initial increase in total cases between May 8 and June 4, followed by two peaks in early June and early July. Although clinical characteristics proved highly variable, the vast majority of patients experienced skin lesions, most commonly located on the genitals, extremities, and perianal region (46%, 39%, and 36% of patients, respectively). The most common non-cutaneous symptoms were fatigue, fever, and headache (59%, 53%, and 51%, respectively).
In an interview with Medscape, Mark Stoové, PhD, chief of public health at the Burnet Institute in Melbourne, Australia, said that while smallpox is not necessarily a sexually transmitted infection, some symptoms of smallpox mirror those of STIs such as syphilis. Therefore, he says, clinicians should be aware of subtle differences, such as the typically painful presentation of smallpox lesions and other nonspecific symptoms that accompany smallpox but not syphilis. He was not involved in the study.
Assembling a response
On May 27, the Quebec Immunisation Committee (CIQ) authorized Modified Vaccinia Ankara-Bavarian Nordic (MVA-BN, Imvamune in Canada; Bavarian Nordic) for prophylaxis up to 14 days after exposure. Because approximately 80% of contacts of infected patients could not be identified or traced, the CIQ in early June authorized the vaccine for PrEP and expanded the indications to include anyone reporting high-risk behaviors in the previous 14 days.
Although MVA-BN is indicated as two doses at least 28 days apart, limited initial supplies necessitated delaying the second dose, except for immunocompromised patients. In June, social venues with on-premises sex accounted for less than 30% of cases. Citywide case totals began to decline around July 10. As MVA-BN supplies increased, second doses became widely available to the target population in October.
“The rapid rollout of the first-dose monkeypox vaccine was key,” says Stoové. Although not noted in the report, he added, the response of Montreal’s gay community to the outbreak was likely also important. “Gay men are generally very health-conscious, especially regarding sexual health. Therefore, the key element in controlling smallpox in Montreal and elsewhere would have been the dissemination of messages through the gay community. This led to high and rapid vaccine uptake and timely recognition of symptoms, presentation, diagnosis, and isolation.”
Throughout the outbreak, only 10 patients required hospitalization. Among the 23,835 people who received the first doses and 1,333 who received the second, 41 were diagnosed with smallpox more than 21 days after vaccination (the maximum incubation period for smallpox).
Vinh acknowledged that occasional breakthrough cases are not surprising, as the vaccine response is unlikely to be uniform. More importantly, he says, Montreal's breakthrough rate is comparable to the 1.4% single-dose breakthrough rate recently reported by the U.S. Centers for Disease Control and Prevention.
Funding to acquire confirmatory test results and genomic data came from the Public Health Agency of Canada. Vinh and Stoové do not report relevant financial relationships.
Ann Int Med. Monkeypox in Montreal: Epidemiology, phylogenomics, and public health response to a major outbreak in North America. Published online December 12, 2022.