Long-acting injectables, innovation, paradigm shift, and quality of life were some of the most frequently mentioned concepts at the launch meeting for Vocabria + Rekambys, the first and only long-acting antiretroviral regimen presented on January 27 and 28. This injectable combination of cabotegravir and long-acting rilpivirine (CAB + RPV AP) was developed to address some of the challenges that still exist regarding the daily intake of antiretroviral therapy (ART) for people living with HIV (PLHIV), including emotional, logistical, and medical issues. A total of 213 healthcare professionals involved in HIV treatment gathered in Málaga to learn about the advancements offered by this new therapeutic option.
The meeting presented the experience acquired in Spain with this injectable antiretroviral regimen during its clinical development (started 8 years ago), since 44 centers have been involved, contributing half of the European patients in the Vocabria + Rekambys registration trials, developed by ViiV Healthcare and Janssen.
Vocabria + Rekambys have been commercially available for administration since December 1, 2022. They are two injectable, long-release nanosuspensions, administered as two intramuscular injections every two months by a healthcare professional.
Este nuevo fármaco representa una innovación en el tratamiento del VIH, y un cambio de paradigma, al ser el primer y único régimen completo de acción prolongada disponible para el tratamiento del VIH.. Está indicado para el tratamiento de la infección por el virus de la inmunodeficiencia humana tipo 1 (VIH-1) en adultos que están virológicamente suprimidos (ARN del VIH-1 <50 copias/ml) en un tratamiento antirretroviral estable, sin evidencia actual o previa de resistencia a, y sin fracaso virológico previo con, inhibidores de la transcriptasa inversa no nucleósidos (ITINN) e inhibidores de la integrasa (INI). EnEspaña se estima que viven 151.387 personas con VIH y que alrededor del 90% de ellas han alcanzado carga viral suprimida. En la última década cada año se diagnostican en torno a 3.500 casos nuevos, entre 8 y 9 al día.
A drug that meets the needs of people with HIV
Needs related to emotional aspects
In recent years, people living with HIV (PLHIV) have raised their voices demanding improvements in their quality of life related to taking daily oral antiretroviral therapy (ART). For a percentage of patients, this still represents a source of stigma, stress, or a constant and unwanted reminder of the infection. Regarding the approval of Vocabria + Rekambys, the European Medicines Agency noted that “for some people living with HIV treated with a stable and effective daily combination of antiretroviral (ARV) medicines, the availability of a long-acting ARV that reduces dosing frequency represents a significant improvement by increasing overall satisfaction with treatment and reducing the burden associated with taking daily pills.”
Data from various studies, both independent and those promoted by ViiV Healthcare, have highlighted the difficulties that taking daily oral ART poses for some people:
- Fear of disclosing their HIV status. The stigma surrounding HIV is a major concern for people living with HIV, often leading them to not disclose their status due to potential social exclusion.
- Anxiety about adherence. People living with HIV are concerned about adherence in order to maintain undetectable viral loads and thus prevent HIV transmission.
- Daily reminder of HIV. Taking daily oral ART has an emotional impact for some people living with HIV, as it is a constant and unwanted reminder of their infection.
Similarly, the Positives Perspectives surveys, which have interviewed 3,497 people in their two editions since 2017, 252 of them in Spain, reveal that more than half of people living with HIV have hidden or concealed their antiretroviral therapy (ART) in the last six months to avoid disclosing their HIV status. Younger individuals, or those with a recent diagnosis, were the most likely to admit to hiding their medication.
Addressing these unmet needs and the challenge of daily ART, the main clinical guidelines for HIV treatment—the American DHHS guidelines and the Spanish GESIDA guidelines—in addition to recommending the use of long-acting ART, recognize for the first time the potential benefit of ART on the quality of life of people living with HIV. According to the GESIDA guidelines, “some of the advantages it offers are: less frequent dosing, reduced stigma or anxiety about disclosing HIV status associated with taking pills, the daily reminder of being HIV-positive, and avoiding the worry of traveling to countries with restrictive laws.”
Dr. Federico Pulido, an internal medicine specialist at the 12 de Octubre Hospital in Madrid, considers the arrival of this medication a paradigm shift, since “the main problem with this infection today is not controlling the virus but avoiding the stigma. With the arrival of long-acting cabotegravir + rilpivirine, once treatment has begun, the patient will receive treatment six times a year, and many may prefer this because it avoids some of the stigma associated with the infection and the daily reminder of it. It remains an infection that many patients want to keep private. This treatment, administered every two months in the hospital, allows them to avoid integrating the treatment of the infection into their daily lives.”
Dr. Eugenia Negredo, an internal medicine specialist at the Germans Trias i Pujol Hospital in Barcelona, agrees that “oral treatments involving a pill every day remind some patients that they need to be taking their medication. That's why intramuscular administration every two months gives this medication a very important added value.”
Dr. Marisa Montes, an internal medicine specialist at La Paz Hospital in Madrid, has participated—as have Drs. Pulido and Negredo—in the clinical development of long-acting cabotegravir + rilpivirine since 2014. She comments that the benefits perceived by patients may lead them to request this treatment themselves, "because it brings to the table elements that clinicians are sometimes not so aware of, such as the impact on quality of life."
Needs related to suboptimal adherence to chronic treatment
As discussed at the presentation meeting, some patients may prefer long-acting cabotegravir plus rilpivirine on their own initiative . Healthcare professionals in Malaga agreed on the need to listen to patients and to consider the unmet needs associated with daily antiretroviral therapy (ART). Furthermore, this new treatment could benefit another group of people living with HIV: those experiencing fatigue from daily ART, recurrent non-adherence to daily oral medication, or, as previously mentioned, those for whom daily ART is a source of stress or a reminder of their illness, among other psychosocial factors.
For Miguel Ángel Calleja, head of pharmacy at the Virgen Macarena hospital in Seville, an appointment every two months to receive injectable medication can allow for more direct monitoring of medication compliance than daily oral ART: “As soon as they miss an appointment, we can notify them, and the dosing window allows us to administer the dose the next day, or two days after their missed appointment.”
Thus, as it is a directly observed treatment (DOT), healthcare professionals have documented medication adherence for patients attending their appointments. In this regard, in the analysis of the LATTE-2 study at 256 weeks of treatment with Vocabria + Rekambys, 96% of patients attended their injection appointment within the flexible dosing window of +/- 7 days.
through daily medication. It remains an infection that many patients prefer to keep private. This treatment, administered every two months in the hospital, allows them to avoid integrating infection management into their daily lives.”
Dr. Eugenia Negredo, an internal medicine specialist at the Germans Trias i Pujol Hospital in Barcelona, agrees that “oral treatments involving a pill every day remind some patients that they need to be taking their medication. That's why intramuscular administration every two months gives this medication a very important added value.”
Dr. Marisa Montes, an internal medicine specialist at La Paz Hospital in Madrid, has participated—as have Drs. Pulido and Negredo—in the clinical development of long-acting cabotegravir + rilpivirine since 2014. She comments that the benefits perceived by patients may lead them to request this treatment themselves, "because it brings to the table elements that clinicians are sometimes not so aware of, such as the impact on quality of life."
Needs related to suboptimal adherence to chronic treatment
As discussed at the presentation meeting, some patients may prefer long-acting cabotegravir plus rilpivirine on their own initiative . Healthcare professionals in Malaga agreed on the need to listen to patients and to consider the unmet needs associated with daily antiretroviral therapy (ART). Furthermore, this new treatment could benefit another group of people living with HIV: those experiencing fatigue from daily ART, recurrent non-adherence to daily oral medication, or, as previously mentioned, those for whom daily ART is a source of stress or a reminder of their illness, among other psychosocial factors.
For Miguel Ángel Calleja, head of pharmacy at the Virgen Macarena hospital in Seville, an appointment every two months to receive injectable medication can allow for more direct monitoring of medication compliance than daily oral ART: “As soon as they miss an appointment, we can notify them, and the dosing window allows us to administer the dose the next day, or two days after their missed appointment.”
Thus, as it is a directly observed treatment (DOT), healthcare professionals have documented medication adherence for patients attending their appointments. In this regard, in the analysis of the LATTE-2 study at 256 weeks of treatment with Vocabria + Rekambys, 96% of patients attended their injection appointment within the flexible dosing window of +/- 7 days.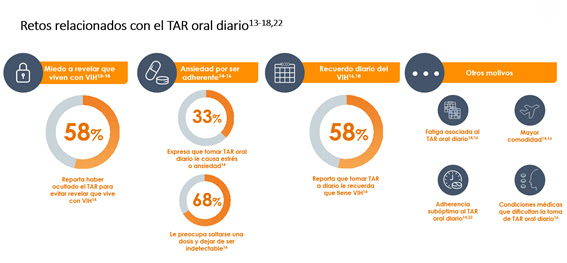
More than eight years of clinical development
Spanish participation in the clinical development of CAB + RVP AP began in Phase II, in 2014, more than eight years ago. Vocabria + Rekambys is being launched after the completion of 9 clinical trials (3 Phase II, 4 Phase III/IIIb, and 2 implementation trials) conducted in more than 25 countries with over 4,000 participants, more than 500 of whom were women.
Another point highlighted at the meeting is the remarkable Spanish participation in this clinical development, from the early stages. In the phase 2b study (LATTE-2), Spain had the highest participation rate worldwide (104 of the 310 participants), and in the registration studies (ATLAS, FLAIR, and ATLAS2-M), half of the European participants were recruited here. In total, Spain has recruited 18% of the patients throughout the drug's clinical development, more than 600 from 44 hospitals. According to Federico Pulido, “the fact that so many Spanish hospitals, doctors, and nurses already have experience with this treatment will allow us to adopt it more easily and adapt it to routine clinical practice more quickly. We have nurses already trained to administer it and doctors who are already familiar with its characteristics and indications.”
One aspect highlighted at the meeting was how the clinical development of Vocabria + Rekambys has been adapted and complemented to provide answers to questions that have arisen over these 8 years, responding to the needs of healthcare professionals and patients.
Furthermore, the various researchers who participated as speakers emphasized that clinical development has gone beyond traditional efficacy and safety analyses to incorporate the patient's perspective on other relevant aspects related to satisfaction, preference, and quality of life. In fact, during the meeting, there were repeated references to semi-structured qualitative interviews and patient-reported outcomes, which have been incorporated as secondary objectives in study protocols throughout the clinical development process.
Efficacy and safety
CAB + RPV AP, administered every 4 weeks in the ATLAS and FLAIR trials, demonstrated non-inferiority to daily oral ART at 48 weeks and this non-inferiority was maintained long-term, up to 124 weeks in FLAIR. CAB + RPV AP administered every 8 weeks in the ATLAS-2M trial demonstrated non-inferiority to administration every 4 weeks at 48 weeks and this was maintained long-term (152 weeks). Long-term confirmed virologic failure (CVF) rates were low, approximately 1.4% (23/1,965), with CAB + RPV AP resistance mutations detected in most CVF cases. A multivariate analysis of long-term viral load factors (VLFs) indicates that the combination of at least two of the following baseline factors may be associated with an increased risk of virological failure: archived rilpivirine resistance mutations, HIV-1 subtype A6/A1, or BMI ≥30 kg/m2. The absence of at least two of these baseline factors predicts the absence of VLFs in 99.1% of cases (negative predictive value).
En cuanto a la seguridad, a 48 semanas tanto en los ensayos ATLAS y FLAIR como en ATLAS-2M el 95% de los efectos adversos fueron leves o moderados, CAB + RV AP presentaron una baja incidencia de eventos adversos graves asociados a fármaco (<1%) y solo un 2% de los participantes discontinuaron por eventos adversos. A largo plazo, hasta 3 años en la fase 3 (ATLAS 2-M) y 5 años en la fase 2 (LATTE-2), Vocabria + Rekambys se siguió tolerando bien, el perfil de seguridad fue consistente con el observado anteriormente, y no se reportaron nuevas señales de seguridad.
Of the participants who received CAB + RPV AP every 2 months, 76% reported injection site reactions, with pain being the most frequent. 98% of injection site reactions were mild or moderate and decreased over time. The median duration was 3 days, and less than 2% of participants discontinued treatment due to injection site reactions.
The most frequently reported adverse reactions in the 2-month dosing in the ATLAS-2M study were injection site reactions (76%), headache (7%) and pyrexia (7%).
The long-term efficacy, safety, and tolerability results of CAB + RPV AP support its use as a complete regimen to maintain virological suppression in virologically suppressed patients.
The meeting included extensive discussion on patient selection for this regimen, for which information is already available regarding baseline factors predictive of virological failure, identified in multivariate analysis with a very high negative predictive value. Along with these baseline factors, which are easily incorporated into clinical practice, the meeting emphasized (as indicated in the Vocabria and Rekambys product information sheets) that before initiating injectable Vocabria + Rekambys, healthcare professionals must carefully select patients who meet the required injection schedule and advise them on the importance of adhering to scheduled dosing visits to help maintain virological suppression and reduce the risk of viral rebound and the potential development of resistance if doses are missed.
The innovation: nanocrystals that gradually release the medication
Vocabria + Rekambys bases its sustained release on aqueous suspensions of nanoparticles or nanocrystals that allow the medication to dissolve and be released gradually after administration by intramuscular injection, maintaining drug concentrations for 2 months, at which point they must be administered again.
How was this prolonged release achieved? CAB + RVP AP They enter the bloodstream at a slower rate than they are eliminated, as a result of slow absorption from the gluteal muscle into the systemic circulation, limited by the dissolution of the nanocrystals.
Implementation clinical trials
During the Vocabria + Rekambys presentation meeting, specialists also addressed the implementation of this new HIV treatment in hospitals. In fact, another important innovation is that, as part of the clinical development of CAB + RVP AP, two implementation clinical trials (CUSTOMIZE and CARISEL) were conducted. While there are no universal formulas, as each center has its own characteristics and specific needs, several models already exist that can be helpful. The main points highlighted at the meeting in this regard were the preparation of the center and the team, emphasizing multidisciplinary coordination to support patients and adapt by integrating the lessons learned throughout the process. Meeting attendees emphasized that the advantages this regimen offers to some patients will encourage centers to find the most efficient way for them to benefit from this new option, which is now a reality.

