Have the authorities made similar mistakes to those made with oseltamivir?
By Michael van den Heuvel
Remdesivir is no longer recommended for hospitalized patients with COVID-19, regardless of disease severity. There is currently no evidence that the drug improves survival rates or reduces the need for ventilation, writes a panel of international experts from the WHO's guideline development group in the BMJ. [ 1 ] Paying a high price for remdesivir without good evidence of lower mortality is like gambling. Dr. Robin Ferner
"Paying a high price for remdesivir without good evidence of lower mortality is like gambling," says Dr. Robin Ferner, a clinical pharmacologist at the University of Birmingham in Birmingham, UK, who was not involved in the publication.
High expectations at the beginning of the SARS-CoV-2 pandemic
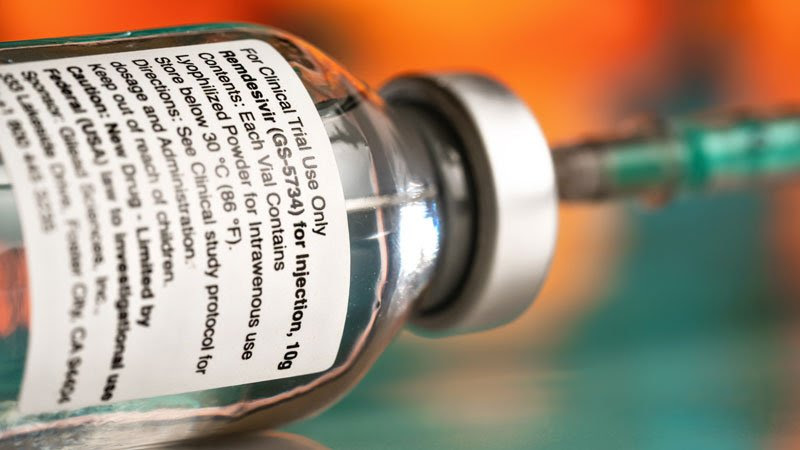
Remdesivir has received worldwide attention as a potentially effective treatment for severely ill COVID-19 patients and is increasingly being used to treat patients in hospitals. However, its relevance in clinical practice is unclear.
As recently as October 2020, the results of the ACTT-1 study indicated that remdesivir had a beneficial effect on morbidity and mortality, but only in certain subgroups (as reported by Medscape ) . According to the European approval , the drug benefits adults and adolescents aged 12 years and older who are receiving supplemental oxygen (therefore, neither those patients requiring ventilation nor those with very mild illness). However, these recommendations could change with the publication of new data.
New data evaluated
The current recommendation is based on four randomized studies with over 7,000 patients hospitalized with COVID-19. These patients received various therapies. The expert panel concluded that remdesivir did not significantly affect mortality or any other important patient endpoints, such as the need for mechanical ventilation or time to clinical improvement of symptoms.
The authors put into perspective that it cannot currently be said that remdesivir is not generally useful. Rather, based on the available data, there is no evidence that it influences important endpoints. Further studies are needed.
Corticosteroid recommendations
The authors also evaluated other therapies. They recommend corticosteroids, primarily dexamethasone (5 mg per day), for 7 to 10 days. Alternatively, clinicians may administer hydrocortisone (50 mg per day), methylprednisolone (10 mg per day), or prednisone (40 mg per day) for 7 to 10 days. Patients with severe to critical COVID-19 benefit from these.
The quality of the evidence is "moderate," the article states. This therapy is not recommended if the disease course is mild. The basis for this is the RECOVERY study, which Medscape has reported on .
Will the story of oseltamivir repeat itself?
In an accompanying editorial, Jeremy Hsu, an American science journalist, explores the question of what the consequences of remdesivir use has in terms of health economics. [ 2 ] He draws parallels with another drug.
“Since the early 2000s, governments have spent billions of dollars stockpiling the antiviral drug oseltamivir ( Tamiflu ) in anticipation of influenza pandemics,” Hsu writes. “Years later, independent researchers gained access to unpublished clinical trials, which showed that the drug has only modest effects on reducing the duration of symptoms, has many side effects, and that there is insufficient data to conclude whether it can prevent the most serious complications of influenza.”
History seems to be repeating itself: “By 2020 at the latest, we will have remdesivir, an experimental and expensive antiviral drug, one of the first and therefore most publicized drugs developed for COVID-19,” the editorialist comments. As is well known, remdesivir traces its origins to joint research projects between the U.S. Centers for Disease Control and Prevention (CDC) and the U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID). They were searching for therapies against the Ebola virus. But the breakthrough did not materialize. Consequently, the hope was all the greater that it could help patients with COVID-19.
However, none of the randomized controlled trials published to date have shown that remdesivir saves significantly more lives than standard medical treatment,” the science journalist summarizes. “Instead, researchers are now focusing on moderately ill patients, who could still benefit from the drug if administered early,” Hsu said. “But what is recommended for remdesivir, given its high price, limited supplies, and now limited usefulness? Should doctors consider using the drug?” Based on the data reviewed, doubts remain.
The question of remdesivir's value in treating COVID-19 can only be answered once the manufacturer, Gilead, publishes all the clinical trial data, as Roche did with Tamiflu in 2013. None of the randomized controlled trials published to date have shown that remdesivir saves significantly more lives than standard medical treatment. Jeremy Hsu
“It was only when we could see everything that we realized Tamiflu ’s advantages consisted solely of shortening the duration of the illness [in the case of influenza] by a few hours,” says Dr. Tom Jefferson of the Cochrane Collaboration . He sued Roche , citing the U.S. False Claims Act. This federal law holds individuals and companies liable if they profit from false statements.
In any case, Hsu hopes history won't repeat itself. He also mentions corticosteroids as a therapy for COVID-19. There is good data, and the drug is inexpensive.
A remdesivir derivative as a new beacon of hope
However, Gilead has another ace up its sleeve: the antiviral molecule GS-441524, a derivative of remdesivir. It is currently being investigated in animal studies and may have some advantages. GS-441524 can be administered in tablet form and can therefore be used earlier than intravenous remdesivir.
Furthermore, its production is significantly cheaper. Remdesivir was the preferred candidate at the beginning of the pandemic because toxicological studies from the Ebola era were already available.
From: https://espanol.medscape.com/verarticulo/5906249