By Paul Sax, HIV and ID Observation
Quick answer: it's complicated.
Let's begin with a clinical anecdote, legitimately considered the weakest form of evidence, but paradoxically it has great power over us because we are imperfect humans.
In April, one of my patients with stable HIV arrived at the hospital with COVID-19 pneumonia. (Some details have been changed for privacy reasons.)
She works as a hairdresser in a community hit hard by the pandemic, where mask use was inconsistent. As soon as she developed a fever, chills, and back pain, she knew what she had.
She must have had symptoms for no more than 36 hours when she arrived at the hospital, and because she has HIV (who knows if this worsened the results?) and is quite overweight, she was admitted.
She enrolled in the SIMPLE trial, which compared remdesivir for 5 or 10 days with the standard of care—all open-label—and was randomly assigned to the 5-day regimen. She received her first dose the night of admission. The next day, she was like a new person. Her fever had broken, she was breathing more easily, and she told me her back pain disappeared as soon as the first dose was completed. She was discharged from the hospital on the third day and made a full recovery. Of course, she could have recovered just as quickly without remdesivir; that's the problem with an anecdote.
But based on this and other cases I saw and the extensive experience of my tireless colleagues, Dr. Francisco Marty and his team, who enrolled dozens of patients in this larger and more rigorous study, published in May, reported a significantly faster recovery in the treatment group than in the controls.
This study, called ACTT-1, now with final results, included a placebo group, providing much stronger evidence that remdesivir actually works. It worked particularly well in people with short symptom durations and in those requiring oxygen. It did not help the sickest people who needed mechanical ventilation or ECMO.
When the ACTT-1 data became available, we created a construct about these critically ill patients who did not benefit from remdesivir. From this perspective, they were in the immune phase of the disease, where the body's immune response to the infection caused more illness than viral replication.
We cannot expect an antiviral to control these processes in the inflammatory phase. Like oseltamivir or baloxavir for influenza, they should be prescribed early, as remdesivir should be when treating SARS-CoV-2. Let dexamethasone or some other immunomodulator do the work later on.
But there were always gaps.
First, the original Chinese study of remdesivir showed no benefit from the treatment. Yes, it had little impact due to the decrease in the number of cases, but the drug also did not reduce the viral load in recipients.
Second, the open-label study in which my patient enrolled had a curious result in the 10-day arm: no apparent benefit compared to the standard 5-day treatment. (The 5-day arm did show benefits.)
How do we explain it?
We now have the provisional results of the SOLIDARITY study, at least in pre-print form and with many more gaps.
In SOLIDARITY, more than 11,000 hospitalized patients with COVID-19 (from 405 hospitals and 30 countries) were randomized between locally available study medications and an open-label control. This included up to five options: four active treatments versus the local standard of care. The medications were lopinavir/ritonavir, hydroxychloroquine, interferon beta-1a, and remdesivir.
(Based on the limited availability of some of the drugs in the countries, the study arms differed in size.)
The results of all interventions showed no survival benefit, and the survival curves for the remdesivir arm versus the standard of care show a slight drop but ultimately remain the same:
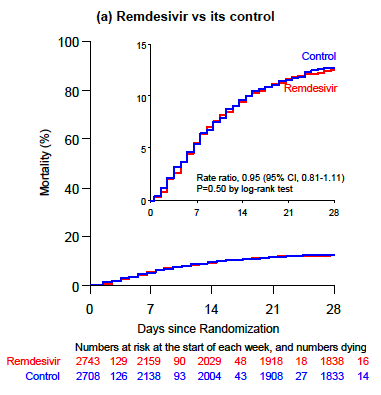
Curves that overlap with such precision are rarely drawn. This image has been seared into the retinas of identification doctors since the preprint was released last week.
So how do we explain these conflicting results? We can't do so completely given the different study designs and populations. But just as the benefits of ACTT-1 cannot negate the results of SOLIDARITY, neither can SOLIDARITY negate ACTT-1 or the 5-day results of SIMPLE.
So let's put all the studies together, as shown here in this colorful meta-analysis, and exclude patients receiving mechanical ventilation, since no study demonstrated benefits in this population:
Mike Johansen
Remdesivir meta-analysis (sensitivity analysis no. 1 / version no. 6)
* This excludes patients on mechanical ventilation (there is one from the Lancet article, 2020 that I could not remove).
Random effects were used.
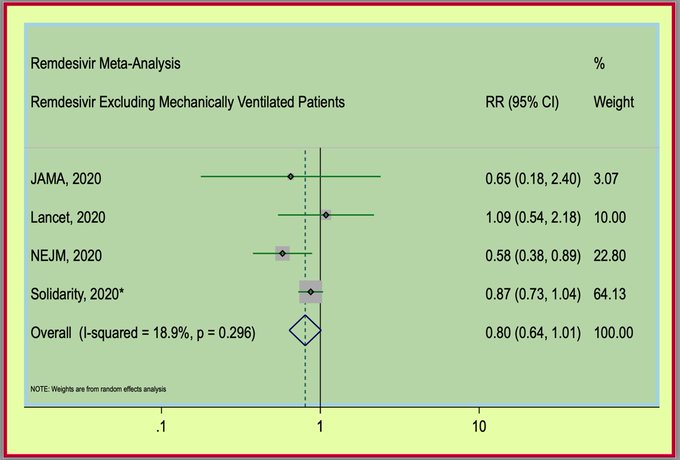
First, we observe that the SOLIDARITY sample size is much larger and exceeds that of the other studies. Second, the point estimate crosses 1 (no benefit), but falls to the left of the line, suggesting some benefit.
If we had to speculate where we would see the greatest benefit from remdesivir, it would be in patients with shorter symptom durations. Even in the Chinese study, those with fewer days of illness fared better than the controls. Could the favorable results in ACTT-1 be due to the fact that 25% of the patients had symptoms lasting less than six days?
In this regard, SOLIDARITY began recruiting patients in March, and for much of the enrollment period, COVID-19 patients did everything possible to avoid hospitalization. For many, the short window of effectiveness for this antiviral had passed by the time of admission. The duration of symptoms, which is not reported in the preprint, is a crucial piece of information.
So, for now, the answer to the question, “Does remdesivir really work?” is sometimes, for some people. Which, given the lack of any other options at this time and its low toxicity, means I would still recommend it for most people hospitalized with COVID-19, hoping to administer it as early as possible, especially for those on oxygen and at high risk of disease progression.
More effective options are needed.

